FAQs
We strongly recommend reading NHS Digital's Practice Level Prescribing in England: a summary, including their excellent More information section, which describes which practices and professionals are included in the data and how prescription data are collected and glossary of terms.
Is your question not listed below? Send us a message here and we'll get back to you
General
How do I download an extract of the data?
How do I cite OpenPrescribing?
What time period does the dataset cover?
How can I access data older than available on your website?
Why do OpenPrescribing figures not always match with other prescribing data analysis platforms?
How do I subscribe/unsubscribe from emails?
Prescribing data
Where do you get the data from?
Is there any patient age or disease information in the dataset?
How do you deal with BNF code changes?
How do you determine ICB membership when GP practices have moved ICBs or ICBs have merged?
Do items claimed on FP34 forms show on OpenPrescribing?
How do items dispensed under a Serious Shortage Protocol (SSP) appear in OpenPrescribing?
How are price concession costs calculated?
Analyse
How do I use the analyse page?
What denominator should I use?
Dashboards
How do I interpret the measures?
How do you calculate savings for ghost-branded generics?
How do I decrease our use of ghost-branded generics?
A GP Practice has closed or merged - why is it included on your dashboards?
Long term trends
How is this different to the data on the rest of the site?
How can I share the view I created?
Disclaimer
General
When do you update the site?
We use the English prescribing dataset from the NHS Business Services Authority (BSA). The BSA have a release calendar for this dataset, although the data is sometimes released early. We aim to update OpenPrescribing by the Wednesday following the release date, but do try to update earlier if the BSA publish sooner. The data is two months behind, so for example January's prescribing data is published in March.
You can find out when OpenPrescribing has been updated by following us on Twitter or signing up to one of our e-mail alerts.
How do I download an extract of the data?
We have written a blog post on how to download data from the Trends and Analyse sections of the site. It is now also possible to download data extracts from our dashboard pages.
How do I cite OpenPrescribing?
You are welcome to use data or graphs from this site in your academic output with attribution. Please cite OpenPrescribing.net, Bennett Institute for Applied Data Science, University of Oxford, 2025 as the source for academic attribution.
If you use data or images from this site online or in a report, please link back to us. Your readers will then be able to see live updates to the data you are interested in, and explore other queries for themselves.
What time period does the dataset cover?
We have data from the past 5 years on OpenPrescribing.net, and from 1998 in the Long Term Trends section.
How can I access data older than available on your website?
Given our limited resources, we do not routinely provide data not accessible through the website. However, you may be able to access this data yourself. The NHS BSA openly publishes the English prescribing dataset dating back to January 2014. If data prior to 2014 is required, the Prescription Cost Analysis dataset is accessible from 1998 onward although is not as detailed as the English Prescribing Dataset.
Why do OpenPrescribing figures not always match with other prescribing data analysis platforms?
At OpenPrescribing we anticipate that our results will not always be exactly the same as those displayed on alternative data analysis platforms. There are two common reasons for this. Firstly, different platforms may use different underlying datasets. At OpenPrescribing we use two openly available datasets: the English Prescribing dataset (EPD) and the Prescription Cost Analysis (PCA). Secondly, different platforms will make different choices in their analytic code for preparing the data and displaying it to users.
All our code used in data preparation, analysis and visualisation is openly available on the Bennett Institute Github for inspection and re-use by anyone. You can also click on the "view technical details for this measure" link in each measure to show exactly what code we are using to create it.
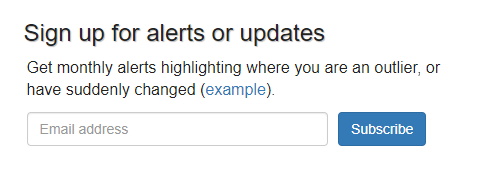
How do I subscribe/unsubscribe from emails?
You can subscribe to our monthly prescribing update emails by going to the dashboard for your chosen organisation (for example by using the "Area and Practice Dashboards" option from our navigation bar), then entering your email address under the "Sign up for alerts or updates" section:
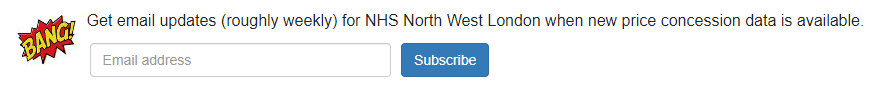
From the organisation dashboard you can also navigate to the price concessions dashboard and subscribe for regular price concession data email alerts:
You can also subscribe to our regular OpenPrescribing newsletter updates
You can unsubscribe or manage your alerts by following the link included at the bottom of all our emails:

Prescribing Dataset
Where do you get the data from?
See our About page.
Is there any patient age or disease information in the dataset?
No, the prescribing dataset we use does not contain any patient information.
What is a prescription item?
Items counts the number of times a medicine has been prescribed. It says nothing about how much of it has been prescribed (for that see quantity) as some presciptions will be for many weeks’ worth of treatment while others will be much smaller.
For more detail see the NHS Digital Practice level prescribing glossary of terms
What does quantity mean?
Quantity is the total amount of a medicine that has been prescribed, but the units used depend on the particular form the medicine is in:
• Where the formulation is tablet, capsule, ampoule, vial etc the quantity will be the number of tablets, capsules, ampoules, vials etc
• Where the formulation is a liquid the quantity will be the number of millilitres
• Where the formulation is a solid form (eg. cream, gel, ointment) the quantity will be the number of grams
• Where the formulation is inhalers the quantity is usually the number of inhalers (but there are occasionally inconsistencies here so exercise caution when analysing this data)
Care must be taken when adding together quantities. Obviously quantities cannot be added across units. But even within a given unit it may not make sense to add together quantities of different preparations with different strengths and formulations.
For more detail see the NHS Digital Practice level prescribing glossary of terms
What is actual cost?
Actual cost is the estimated cost to the NHS of supplying a medicine. The Drug Tariff and other price lists specify a Net Ingredient Cost (NIC) for a drug, but pharmacists usually receive a discount on this price. Additionally they receive a "container allowance" each time they dispense a prescription. The actual cost is estimated from the net ingredient cost by subtracting the average percentage discount received by pharmacists in the previous month and adding in the cost of the container allowance.
For more detail see the NHS Digital Practice level prescribing glossary of terms
How do you deal with BNF code changes?
Chemicals can occasionally be re-classified from one BNF code to another. An example of this is Linaclotide, which changed from 0102000AH to 0106070B0 in 2013.
We have aimed to normalise the data so that past prescribing is always listed under the current BNF code. This means if you look at Linaclotide’s trends page, you will see the prescribing pre-2013 even though the code changed.
We have done this by mapping the old codes to the new codes, by using a map the NHS Business Services Authority provided us in a personal communication (after correcting for obvious typos etc.). You can read how we did this on github.
You can read more about BNF codes and how they are structured here.
How do you determine ICB membership when GP practices have moved ICB or ICB have merged?
We use the current Integrated Care Board (ICB) membership of a practice in our analyses. If a GP Practice moved ICB, the data from before the move will be included in the data for their current ICB. Their former ICB will not include that practice's data. This means that when you look at our prescribing measures, you will be looking at the past prescribing of the current GP practices in that ICB.
When ICBs merge, we create a new dashboard for the newly formed ICB and remove the old ICB dashboards. We use the prescribing data for the current practices in the new ICB in our measures. Any data for practices in the old ICBs that closed prior to the merger will be included in the new ICB's data
Data from our analyse and trends pages also use current practice membership of a ICB when aggregating the data by ICB
Do items claimed on FP34 forms show on OpenPrescribing?
Medicines claimed under an "FP34" form by GP practices fall into two categories:
- High Volume Vaccines (influenza, typhoid, hepatitis A, hepatitis B , pneumococcal, meningococcal vaccines). These vaccines are purchased by the practices, and are claimed for on FP34D (for dispensing practices) and FP34PD (for other practices) appendices. No FP10 is required to claim these items.
- Other items (for example injections and local anaesthetics. These items are often "personally adminstered" by GP practices, and although the payment for the administration is claimed for via the FP34 form by the practice, a separate FP10 for item will need to be submitted as well.
Medicines administered to patients under both categories appear in OpenPrescribing data. For example here is data on influenza vaccines, which are usually claimed on FP34D/PD.
How do items dispensed under a Serious Shortage Protocol (SSP) appear in OpenPrescribing?
The BSA have confirmed that the prescribing dataset we use shows entries for items dispensed under an SSP, but does not include information on the item originally prescribed.
How are price concession costs calculated?
The extra costs for an organisation implied by a price concession are calculated as the difference between the drug tariff price and the concession price after applying the relevant discounts, multiplied by the quantity prescribed by that organisation.
For concessions prior to April 2023 we appply the national average discount of 7.2% to both the tariff price and the concession price. From April 2023 onwards we no longer apply any discount to the concession price, as specified by CPE. From April 2024, we apply a different discount rate to tariff prices depending on the category of the product: 20% for generics, 9.85% discount for appliances and 5% discount for everything else.
Analyse
How do I use the analyse page?
Instructions are found here.
What denominator should I use?
We use this site a lot for our own work and our own research. When we use the analysis page, we find it useful to choose denominators cautiously.
You can use "list size" which tells you how many patients a practice covers, but this can be problematic, because different practices will have different kinds of patients, some with lots of older people, and so on.
When using the data ourselves we tend to use more thoughtful approaches to try to "bake in" population prevalence or need for a particular condition, or to explore different prescribing patterns. For example, we often use whole classes of drug as the denominator in our analyses, as in the video walkthroughs; or we compare the use of one drug against the use of another. When looking at whether a practice is using a lot of Nexium (an expensive "proton pump inhibitor" pill for treating ulcers) we might look at "Nexium prescribing" versus "all proton pump inhibitor prescribing" (example).
We have removed STAR-PUs as a denominator option on our analysis page and from several of our pre-built antibiotic measures. This decision was made following user consultation, due to concerns about the appropriateness of the STAR-PU weightings, which have not been reviewed since 2013. You can read more about this in our blog.
Play around and let us know if you find anything interesting, or develop any interesting methods.
Dashboards
How should I interpret the measures?
We recommend reading NHS Digitals's Practice Level Prescribing in England: a summary, including the more information section and glossary of terms.
Just because a practice or NHS organisation is an outlier for high or low use of a particular treatment, that doesn't necessarily mean they are good or bad prescribers. These are measures rather than indicators, and they need to be interpreted judiciously.
For example, a practice that prescribes a lot of benzodiazepines may have a lower threshold for prescribing them, or may run a specialist service for people with substance misuse, or have one doctor with an interest in - and long list of - such patients.
How are the measures chosen?
Our original measures were developed in discussion between Drs Jeff Aronson, Kamal Mahtani, Ben Goldacre at the University of Oxford, and Richard Croker from NHS Devon. The Bennett Institute team now create measures based on ideas for research, national guidelines, and from our users. Please do get in touch if you have an idea for a possible new measure.
How do you work out savings on individual presentations?
There can be huge variation in the price a practice or NHS organisation pay for a treatment, even for the same drug at the same dose. It is well known that branded and generic versions of the same treatment will have different prices; but different specific “brands” of “branded generic” may also have different prices; and there are many other similar sources of variation in price. Every month we estimate what could be saved if every organisation were prescribing as well as the best 10%, for each item prescribed. Read our detailed FAQ on the subject here.
How do you calculate savings for ghost-branded generics?
Ghost-branded generics are generic items which have unintentionally been prescribed with a manufacturer name. For example, Naratriptan 2.5mg Tablets is the correct generic name; a ghost-branded version is Naratriptan 2.5mg Tablets (Teva UK Limited). When an item is prescribed generically, the dispenser is reimbursed at the price in the Drug Tariff; but when a manufacturer is stated, the reimbursement price is usually more expensive.
In some cases, generics will be deliberately prescribed with the manufacturer name, for example where a patient needs to have the same colour pill consistently, or in items with a narrow therapeutic index. However, we assume these are a tiny majority of the total ghost-branded prescribing that happens.
The prescribing data released by NHS BSA aggregates all generic and ghost-generic prescribing together, so it is not possible for us to break down the data precisely.
In addition, the published Drug Tariff prices are not necessarily the ones used by BSA for reimbursement (see our technical notes here for an explanation of this). Therefore, to estimate the size of the problem for individual practices or NHS organisations, we first have to estimate the Drug Tariff price for a generic, which we do by taking the median price paid for that generic item across the entire country. This gives us a price that should be paid for all prescriptions of that generic. We then compare this with the price that was used for reimbursement. This difference gives a reasonable estimate of the total possible savings if a generic were prescribed correctly. We only include savings of more than £5. Note that we use Net Ingredient Cost for our calculations; the true reimbursement cost will typically be 7% less due to bulk discounts.
You can read more about the background of this issue on our blog.
How can I decrease our use of ghost-branded generics?
The spreadsheet download on ghost-branded generics page is sorted by total possible savings. The practices and items appearing near the top of this list are likely to be prescribing the highest level of ghost-branded generics. ePACT2 now carries data at an AMPP level for ghost-generics (termed "premium price generics"), meaning you can identify individual scripts which are at fault; PrescQIPP has also added ghost-generic reports and searches.
In general, the fix will be dependent on your prescribing software. It should be possible to configure it so only true generics can be picked, at least for the most egregious items.
We are currently gathering feedback from practices who have implemented such fixes, and will publish a summary on our blog in due course
.A GP Practice has closed or merged - why is it included on your dashboards?
OpenPrescribing uses the prescribing dataset published monthly by NHS Digital, which includes the past prescribing for practices that have merged or closed. Data will still show for a closed practice if a prescription prescribed by that surgery was dispensed in the community after the closure date. The historical data for closed practices will continue to be shown on OpenPrescribing, and any practice with a current status of Dormant or Closed will be labelled as such. They will continue to be shown under the ICB they were last recorded under.
Long term trends
How is this different to the data on the rest of the site?
This tool uses Prescription Cost Analysis (PCA) data back to 1998, which shows all products dispensed in the community (or personally administered by doctors) in England, whereas the data used elsewhere on the site gives more detail about how items were prescribed (but still only includes those which were dispensed). This causes two main differences to arise:
- generic drug names/codes do not appear in our long-term trends tool until the generic product is available to be dispensed, even if doctors are prescribing generically
- cross-border issues: in our long-term trends tool, items are included which were prescribed by doctors in other parts of the UK, provided they were dispensed in England, but any items are excluded which were prescribed in England and dispensed in other parts of the UK
This data cannot be split down to individual months or practices.
How was the data normalised?
All figures are shown per 1000 population (adjusted for England's mid-year population size each year) and costs are corrected for inflation (using consumer price index), both obtained from the ONS. We also normalised the data by drug name and classification, where possible, such that drugs moving around Chapters, or changing name, do not disrupt the trends. We did this by mapping all drug names to their current position in the BNF. Drug names not matched exactly to a currently available item were assigned appropriate classifications via approximate matching, ensuring that drugs were not inappropriately moved across categories, e.g. those with multiple uses. Some chemicals which are no longer available will not have been assigned an up-to-date chemical code, but will have a chemical name (as supplied in the original dataset) and a product name (derived from the drug name). Efforts were concentrated on Chapters 1-6 and 10 so other Chapters may have more inconsistencies, particularly Chapters 9 (Nutrition) and 13 (Skin). The details are explained further in our paper, currently under review with BMJ Open, and in the code can be found here.
How can I share the view I created?
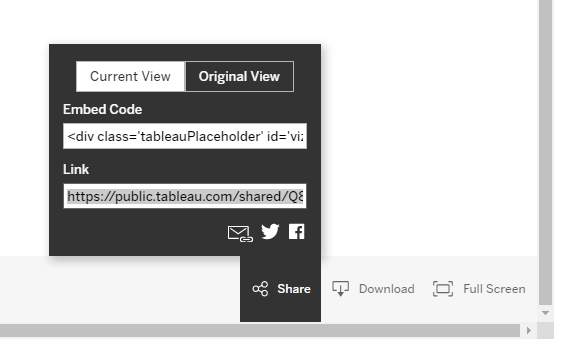
Scroll to the bottom right, then:
- To share the dashboard as a link for someone else to access, or to save for later, select Share. If you have made changes then "Current View" will be selected. Copy the link:
- To share the view as an image or PDF, select Download and select your preferred option.
Can I download the data?
You can download the entire dataset, either the compiled original data or our normalised data, from FigShare.
When will it be updated?
The annual data is released in March each year; however, ONS figures for population size are not available until June.
Disclaimer
We accept no liability for any errors in the data or its publication here: use this data at your own risk. You should not use this data to make individual prescribing decisions.